AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
So2 molecular geometry4/10/2023 119.32xb0 and two equal S-0 bond lengths of 0.143 nm (1). Sulfur dioxide is an angular molecule with a bond angle of. However the bond angle decreases when the electronegativities of ligand atoms are more than that of central atom. I) The bond angle decreases due to the presence of lone pairs, which cause more repulsion on the bond pairs and as a result the bond pairs tend to come closer. So, SO2 has larger bond angle What causes smaller bond angles? So due to more lonepair bond pair repulsion,the bond angle of H2S and H2o is less than that of SO2. Secondly, the compound SO2 contains one lone pair whereas H2O and H2S contain two lone pairs. Does co2 have a bond angle of 120?īoron trifluoride has a bond angle of 120 degrees. these predictions, the bond angles decrease from 116xb0 for OCH2, to 111xb0 for 0CC12, to 108xb0 for OCF2 (33). 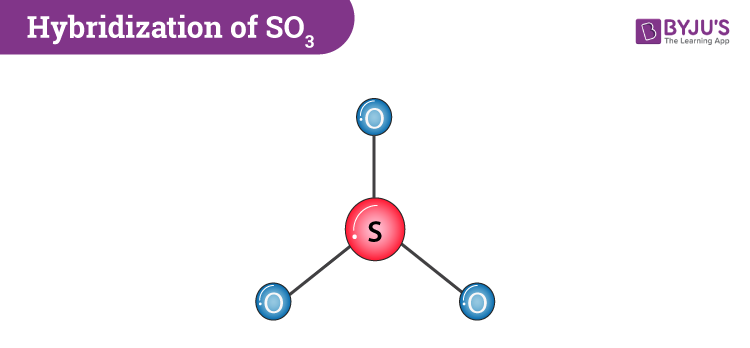 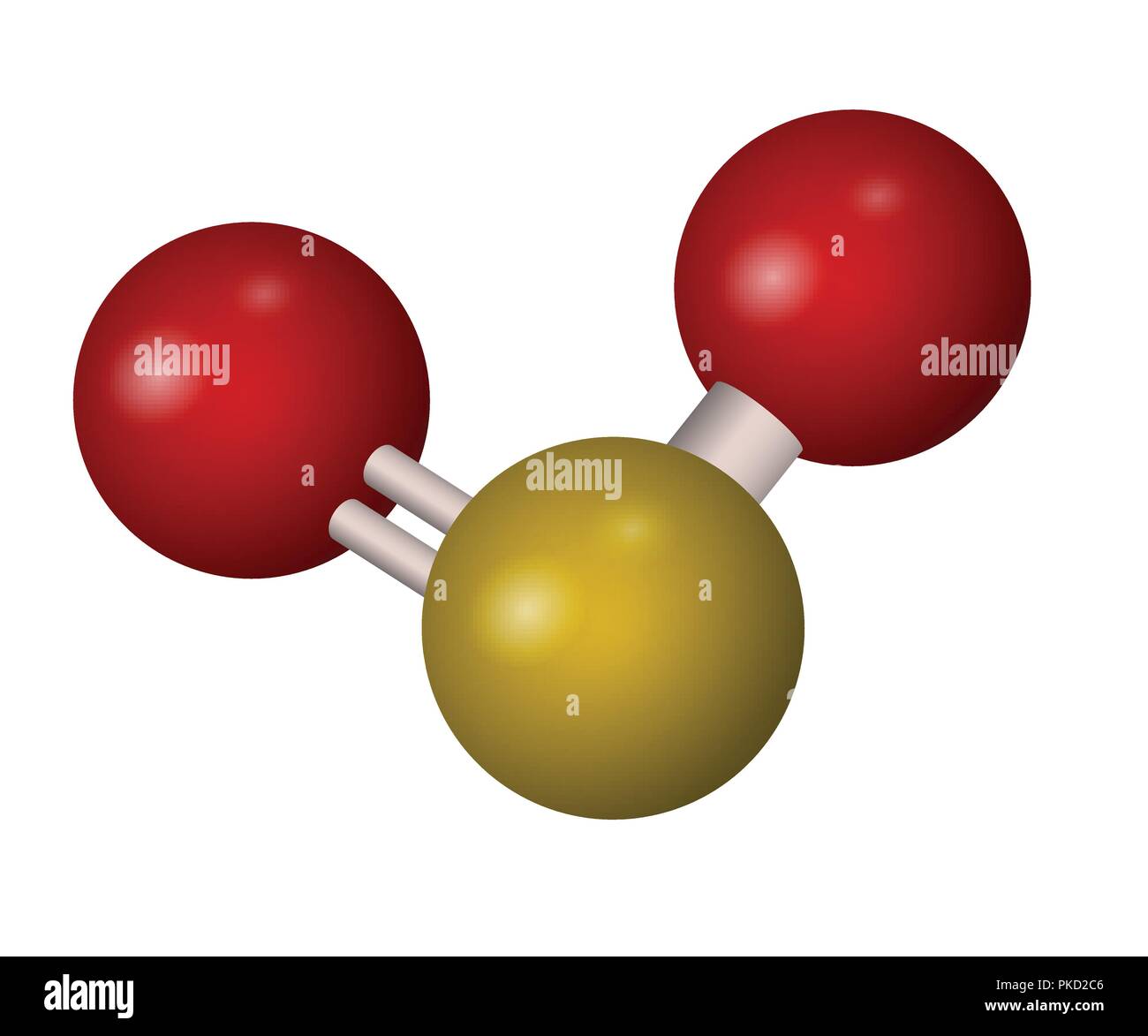
bonding description will explain adequately the observed molecular geometries of both sulfur dioxide and ozone. What has a bond angle of 120?īond angle in sulfur dioxide provides a clear answer. Molecular structure and bonding The molecule SO3 is trigonal planar. Name of moleculeSulfur dioxideBond Angles 119 degreesMolecular Geometry of SO2Trigonal planarHybridization of SO2sp2 hybridizationNo of Valence Electrons in the molecule181 more row Which has more bond angle SO2 or SO3? Note that the precise angle is 106.0 degrees). What is the bond angle of SO3 2?Ī quick explanation of the molecular geometry of SO32- including a description of the SO32- bond angles. Which of the following structures will have a bond angle of 120 degrees around the central atom?īCl3 is sp2 hydribised and is trigonal planar in shape and hence the bond angle is 120u2218. 
What is the bond angle of SO2?īond angle in sulfur dioxide provides a clear answer. Tetrahedral molecules (methane) will have bond angles of about 109 degrees. Trigonal planar molecules (BF3) will have bond angles of 120 because each of the F molecules is spread out on a plane equidistant from each other. These areas also repel the bonds to the maximum distance apart, which is in this case 120 degrees. In SO2 the central sulfur atom is surrounded by three areas of electron density, two bonding, one lone pair. What is the bond angle between SO2?ġ19^u2218 Why is the bond angle 120 degrees? So its basic angle is 120 degrees but the lone pair will squeeze the OSO angle to a bit less than 120. has three domains, two double bonds and a lone pair. If a molecule has two bonding groups and one lone pair electrons, the bond angle will obviously be less than 120 degree and cause a bend molecular shape. Molecules that contain a lone pair on the central atom will cause repulsion and that is the reason. Therefore, the H-O-H bond angle in water is smaller than the O-S-O bond angle in sulfur dioxide. The central oxygen atom of water has 2 non-bonding pairs, compared to the 1 non-bonding pair on the central sulfur atom in sulfur dioxide. It has a bond angle of 120o Does SO2 have a bond angle of 120? SO3 Molecular Geometry And Bond Angles Sulphur trioxide is also non-polar. The extra repulsion of the lone pair and double bonds accounts for the reduction to 119xb0 Why is SO2 120 degrees? 
There are three electron regions so the bond angle is 120xb0. Why is the bond angle of SO2 less than 120?
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
